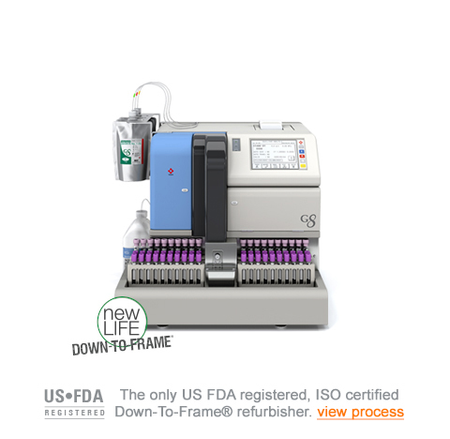
Tosoh G8 HPLC Analyzer
Item# 0-TS-G8
The Tosoh G8 HPLC Analyzer, provided by Tosoh Corporation
Equipment Support![]() Service Options
Service Options![]() Consumables
Consumables![]() Quality Controls
Quality Controls![]() Service Parts
Service Parts
Package Size: EA
Related Products
Diamond Diagnostics presents the refurbished Tosoh G8, an HbA1c analyzer available in a benchtop configuration, manufactured by Tosoh Corporation. With the capacity to process up to 37 tests per hour and featuring a load-up capacity of 90 samples, this analyzer is designed for efficient and accurate analysis of glycated hemoglobin levels. Equipped with advanced technology, the Tosoh G8 delivers comprehensive insights into HbA1c levels, crucial for monitoring diabetes management. Its precision and reliability make it an indispensable tool in healthcare settings, providing valuable information for diagnosing and managing diabetes. Whether in hospital laboratories, clinics, or research facilities, the Tosoh G8 plays a crucial role in patient care by facilitating timely and accurate diagnostic results. By furnishing healthcare professionals with actionable data, it enables informed decision-making and personalized treatment strategies, ultimately contributing to improved patient outcomes and overall wellness. From routine screenings to specialized diagnostics, the Tosoh G8 stands as a trusted ally in the pursuit of better healthcare, offering consistent performance and dependable results in HbA1c testing.
Down-To-Frame Refurbishment Process


Upgrading or Adding Equipment to your lab?
Save up to 70% with refurbished equipment from Abbott, Alfa Wasserman, Beckman, Biomerieux, Biotechnia, Hitachi, Horiba ABX, IL, J&J, Radiometer, Randox, Roche, Siemens, Stago, Sysmex, Thermo and Tosoh.
Diamond is the only USA FDA registered, ISO Certified and Down-To-Frame® refurbisher of Chemistry, Immunology, Hematology, Caogulation and Blood Gas equipment. Inquire today for pricing and Details.
Every analyzer undergoes a systemized, rigorous and unique Down-To-Frame® Refurbishment process to rebuild each analyzer to its original specification and intended use.
Disassembly & Decommission
- System decommissioned from laboratory and decontaminated
- Preliminary inspection of functionality
- System disassembled Down-To-Frame®
Aesthetic Restoration
- All outer covers, chassis and internal surfaces stripped, sand blasted and prepared for painting
- All prepared surfaces primed and painted with 3 part compound, color matched automotive quality paint
- All original logos and signage are replaced in original appearance
Refurbishment & Reassembly
- All mechanical, pneumatic, electronic assemblies disassembled, cleaned, restored, verified and reassembled, replaced where needed
- Disposable components replaced with new components
- New components include: tubing, fittings, syringes, seals, check valves, probes, reagent lines, waste assemblies and similar items
- System completely reassembled into its original condition in accordance to protocol and established specifications
Diagnostics & Troubleshooting
- Assembly Functions & System errors assessed, troubleshooting and repair conducted
- Full system diagnostic testing conducted
- System successfully calibrated with certified calibrator materials to ensure proper functionality
Performance Validation
- Rigorous Multi-Level Quality Control testing conducted on established parameters using certified control material
- Specific parameter testing available upon request
- System checked for power stability, software revisions and other upgrades
Final Packing
- Required peripherals for operation refurbished or provided new as needed
- System properly flushed, drained, protected and prepared for storage
- System professionally crated with heat-treated and non-coniferous wood for international transit
Performance & Configuration
Assays
0 Total Assays Tested
Technical Specifications
| Manufacturer: | Tosoh Corporation |
| Series: | G8 |
| Maximum Throughput: | 37 tests/hour |
| Sample Cycle Time: | 96 (sec) |
| Sample Size Min: | 4000 μL |
| Direct Sample (STAT): | No |
| Auto Sample Handling: | No |
| On-Board Refrigeration: | No |
| Configuration: | Bench Top |
| Height: | 49 cm (19 in) |
| Width: | 53 cm (21 in) |
| Depth: | 52 cm (20 in) |
| Weight: | 34 kg (75 lb) |
| Water Requirement: | No |
| Power Supply: | 110V-220V |
| Point Of Care: | No |
All units sold by Diamond Diagnostics or units qualified by Diamond Diagnostics are eligible for Field Service coverage. Service plans are the best way to maximize your instrument investment and avoid the high costs associated with instrument failure. Our plans are designed to support optimum instrument performance, ensure reproducible results and minimize down-time. Our service plans provide a timely response from our dedicated engineers who are certified on an ongoing basis to ensure top-notch results, even in the most complex laboratory environments.
: Included
: Chargeable
: None
* Contact for more details

Preventive Maintenance
For the laboratory looking to manage manufacture recommended preventative maintenance (PM) schedules for instrumentation at a fixed cost. The PM Service Plan will increase the reliability and reduce the risk and costs of unplanned maintenance and equipment downtime. Our plans are scheduled and planned around your specific instrument and operational needs.

Depot Repair Per Diem
For the laboratory looking to maximize the life of their instrumentation while effectively managing repair costs. The Depot Repair is a per diem plan designed for any size instrument needing major repair, complete rebuild or recycling.

Annual Depot Repair
For the laboratory looking to maximize the life of their instrumentation while effectively managing repair costs. Annual Depot Repair is an annual based plan designed for small to mid-sized instruments needing maintenance or repairs and can be easily shipped by your local carrier to our Depot Service Facility in Holliston, MA.

Phone Support
For the laboratory employing qualified service professionals, the Virtual Plan may be a suitable choice to replace or supplement Field Services. The Virtual plan offers its customers support by email and telephone, within business hours with communication directly with Clinical Engineering staff specialized in the model of instrumentation that the laboratory is utilizing. The Virtual Plan is annual.
Click here to learn more.
The names and logos of manufacturers, their instruments, and their products referred to herein may be protected by trademark or other law, and are used herein solely for purpose of reference. Products are available for international distribution only unless otherwise indicated. Diamond Diagnostics expressly disclaims any affiliation with products it does not manufacture, as well as sponsorship by other manufacturers. For current regulatory status on products within this website, please contact your sales representative. Prices and Information shown are for reference only and may change without notice. SmartLyte®, ProLyte®, CareLyte®, and Down-To-Frame® Refurbishing are Registered Trademarks of Diamond Diagnostics®. No mobile information will be shared with third parties/affiliates for marketing/promotional purposes. All the above categories exclude text messaging originator opt-in data and consent; this information will not be shared with any third parties.